How to draw Lewis structure of any compound? Easy Trick
ฝัง
- เผยแพร่เมื่อ 15 ธ.ค. 2024
- This lecture is about how to draw Lewis structure easily. In this animated lecture, I will teach you drawing Lewis structure of different compounds.
To learn more about how to draw Lewis structure, watch this lecture till the end.
#lewisstructure
#tricktodrawlewisstructure
#chemistry
Subscribe my channel at: / @najamacademy
TH-cam link: / @najamacademy
Facebook link: / najamacademy


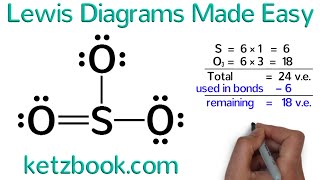






any neet aspirants 😂
Haha here
😂😂
Hello boii😂
Hey bro😅
Jee aspirant 😂😂
bro your explanation kinda kills my fear for my exam , litterally wtf is this , you are amazinggggg❤🔥
More information in less time
Best best best video❤
Supirii bosa cupidiytm theruna😎
Your explanation is just amazing 🤩
Any 2025 jee aspirants😂
Sir, Your short videos make the concepts easy and crystal clear in very less time. Awesome.
Hii
Hi
Much appreciated goes to Najam Academic ,so that you always do great thing to everyone in the world.
Thank you very much Sir. You're explanations are spot on👌
Lots of love from Dubai....✨♾️ 🇯🇵
Thanks sir ap na is structure ka dots samjha dia,love from Pakistan 🇵🇰👍🏼
Waha pr bhi NCERT h Ky 🤡
Tum Padhte bhi ho mujhe laga bas bakwaas krte ho 😂
Better effort🎉
Nice explain
I always like ur videos although i watch it or not because u are just a awesome teacher , hats off to you sir because of you my doubts are cleared very easily within few minutes .Your teaching way is super crazy ❤️❤️🔥🔥 Thank-you Sir
Thank you so much
Explanation with animation..... ❤❤
Please Make video on rutherford atomic model,Experiment,conclusion,defects almost everything about this.please .........
Also about Bohr model.you are best teacher.I am your regular viewer.please .....
Bna rekhi he
Sir jiiii...... Any trick to remember the priority series of functional groups in GOC?
You are just like me
@@filminsights yes. But uska reply nhi aaiga ab kabhi
@@GOATS719 watch pankaj sir lectures....
@@GOATS719 sad reality☹️🥺
Keep practicing questions , at a time it'll be at the tip of your tongue.
You teach very well ❤
Very nice👍👍👍👍
Really nice video and understanding each and every point
Ur technique to teach is amazing
Everything is in chemistry if you know the periodic table perfectly 😓
Thank you sir no waste of time like other youtubers😊
Hii
Insta use karti ho
WOW larki dekhi nhi k setting start
5:48 arrow position for H2O is incorrect
this explanation was too good thank you soo much sir
Sir your voice is cute snd speaking stayle to good these video is really helffull do the studay and concept clearing video
Thank you so much sir 🙂🙂🙂
Thank you so much sir Najam! Your videos are really helpful for students starting to learn chemistry like me 🥰🥰 will surely recommend your channel to my colleagues!
It's my pleasure
❤
very helpful
Sir , please make video on hybridization and shape of molecules 🥺🙏😭
Your videos are amazing and easy to understand.
Bro Hybridization Is So Easy..
@@shivamnewarts291 how , i find it so hard
@@Googleaccounty th-cam.com/video/nNkw_0c8vY0/w-d-xo.html
@@shivamnewarts291 pata nahi kaha say laga ha easy
So hard
Sir you have taken very basic compunds to make the structures what if i have to draw structures like kmno4 or H3PO4 etc
Thank you❤❤❤❤❤❤❤❤
Thanks🎉🎉
Great👍😁
Thanks man❤
Sir very best explanation ☺️
Sir please upload video on "WHAT IS CHARGE"🙏🙏
Good informative vidio😊
Tq sir for the best explanation sir please keep maths also sir please 🥺🥺
Sir please make video on molecular formula
Wow
Thanku so much sir❤️
Concept understood .clearly thankyou
Thank you for the explanation, but can you explain how to determine the placement of elements when drawing the lewis structure? Ex: NH3, does the H element have to be placed as you displayed, or can an H go above the N?
if h goes above the n put your lone pairs bellw
hlw
Thank you so much sir 🙏🙏very helpful ❤️👍🏻👍🏻👍🏻
Thanks for this video.
Its help a lot..❤
This video is very helpful for me thank you sir ji God bless you 🥰🎈🙏🙏❤️💫
Legend trick
thanks 👍 a lot
Sir plz do a video for resonance structures
you deserve an award!
Sir your channel is very good.
Sir make video on main branches of chemistry
SIR 91K SUBS MUBARAK HU 🔥🔥🔥🔥
3:59 nyc explanation 😅😅😅
Tomorrow is exam
Super mind blowing amazing sir❤❤❤
Thanks alot i now understand
Best explanation ❤
Kon kon exam sa phela yea dakhera ha legend student 😎😎
Mee
Lot of thanks
U r really superb
Thnk youu soo much sir💓
Great explain thank you ❤
Thank you so much 💕
Who are u
@@legend07132 I'm your mom 😂🤣
@@konmonidas2695 🤨
@@konmonidas2695 his dad was too lucky that night
I love u u helped me always
Thank you so much sir Najam❤❤❤. God bless you. Your explanation is really amazing
Thank you 😊❤️❤️ so much sir because of you first time I understand this concept just in 6 minute
amazing
Omg finally i understood it thanks a lot!
Explain about acidic nature please
His accent ☠️☠️
👏👏👏👏👏👏👏👏👏
1:21 why we have to put dot on the upper side of H we could have put that in the lower or anywhere else...??
We can put it anywhere
Happy teachers day sir
Thank you
Any neet aspirants 🔥🔥🔥
Excellent explanation
Nice explain thx sir
1:22 -It❌ Ate✅
U focus on the video not the spelling mistake 😂
Thanks Sir 🙏
Thanks thala🎉
What ever u told was very easy try some hard questions najam
Any neet aspirants
Yes... ❤
Thank you sm, it really helped me❤❤
assalamualaikum. thanks for your valuable videos, sir. would you make a structural video on NH4, CO3 HCO3, SO4 PH4 NO2 NO3 SO3 Ion. that means I am requesting a video on Radicals structure
Please do all the concepts in class 11
Hi,your all lectures are fantastic and easy to understand you have cleared my all concept,Thankyou very Much ...Sir how you make this video from which app or software and also thumbnail Pls tell Sir.
Sir g please complete all courses of Ist year
Hi, what software/app do you use to make your videos?
Premium Nokapod software
Thank You Sir❤
Thank you very much sir
u got a new subscriber
10 th class
Making short videos won't be of any help for jee or neet aspirants.....it is a very vast topic....facts
Thankyou so much❤
Bro everyone is mad glazing in the comment section
Watching half hour before exam
My guru