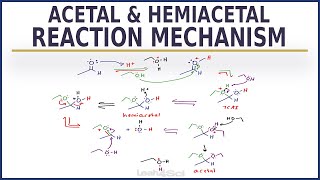
Acetal Ketal Hemiacetal Hemiketal Reaction Overview and Shortcut
ฝัง
- เผยแพร่เมื่อ 11 ก.ย. 2024
- leah4sci.com/ac... Presents: Acetal, Ketal, Hemiacetal, & Hemiketal Reactions Overview and Shortcut in Organic Chemistry
Need help with Orgo? Download my free guide ’10 Secrets to Acing Organic Chemistry’ HERE: leah4sci.com/or...
In this video:
[0:17] Steps for Acetal/Ketal Formation
[2:22] Creation of HemiAcetals and HemiKetals
[3:50] Reversible Reaction and Identification Trick
[6:12] Identifying Reactants and Products
[6:48] Acetal Shortcut
[7:52] Determining Reactants Using a Shortcut
[9:17] Forming a Protecting Group or Gas mask
This video explains the differences in Acetals vs Hemiacetals, and Ketals vs Hemiketals. You'll see an overview and a super simple shortcut to find the product or reactant for an acetal reaction including cyclic acetal protecting group.
Catch the entire Acetal Video Series along with the Acetal Practice Quiz and Cheat Sheet on my website at leah4sci.com/ac...
For more in-depth review on Acetals including practice problems and explanations, come join my online membership site the organic chemistry study hall: leah4sci.com/join
For private online tutoring visit my website: leah4sci.com/or...
Finally, for questions and comments, find me on social media here:
Facebook: / leah4sci
Twitter: / leah4sci
Instagram: / leah4sci
Google+ : plus.google.co...
Pinterest: / leah4sci








You made me understand what I struggled with for weeks during my lectures, in only 12 minutes.
Thank you
Wow, that's awesome! So glad it helped clear things up for you!
This video made so much sense. Everything that made no sense in lecture you explained in 12 minutes. You're the best, Leah!!! Thank you!
I'm so happy I helped you understand, that's always my goal!
@@Leah4sci❤
Thank you so so much, I had no idea what was happening in my Organic classes
I'm glad I could help
You're a life saver thank you so much!!
Happy to help!
Your's way of teaching organic chemistry is awesome thanks a lot a mam
aww... thank you for the kind words
thank you leah i have been watching your videos since 0rgo one you are really an amazing organic teacher
Glad I could help!
Thank u so much, Ms. Leah. You've been helping me a lot.
I'm so glad!
This is the best instructional video of all time
Yay! So glad you like it!
First time understood what acetal and ketal is
I'm so happy to hear this! Thanks for watching.
For organic chemistry I directly search your videos..!!
Awesome!!!! Thanks for that!
Best video to quickly revise at 1.25, thank you 🙏
Glad it was helpful!
Always appreciated for free education, god bless you
You are very welcome!
I am glad all your videos exist !!
I'm glad they do, too!
Thank you I was very confused and u made my concept clear and you told exactly the point I were searching for
You're welcome! Glad to hear it!
Thank you!!! I have been waiting for this for so long!!!!
You are very welcome!!
Thank you Bhat!
Thank you so much for an easy visual breakdown! :)
You're very welcome!
Thanks for the video it’s really help me in my organic 1 class
Glad to hear that!
Yes keep uploading new videos for us I really appreciate it. I’m watching from The Gambia 🇬🇲 it’s in west Africa.
great video. I always felt intimidated any time i heard acetal or ketal. This video breaks it down very simply! THANKS!
Always happy to help! You're so welcome.
@@Leah4sci Do you do MCAT tutoring?
Thank you so much! I was having trouble with this, but everything makes perfect sense now!
Great to hear it helped you!!
I feel like sending you a box of chocolates because you helped me so much to understand this chapter. I having struggle with it for a full week until I found this video. I totally get it now. Thank you so so much.
Yum! I love chocolate :) I'm glad I was able to help!
11:05 Gas mask analogy. Instantly goes from short term memory to hippocampus storage within 5 seconds. Thank you!
WOOHOO! You're welcome!
I always forget this and have to look it up around this part of the quarter. Thanks.
You're welcome!
Wow.your technics are amazing.thank you so much.
You are so welcome!
Thank you. Short and sweet. Very helpful review.
You're very welcome, glad you found it helpful.
Thanks for taking effort to make this vedios and making it very simple
You're very welcome!
Thank you!!!
You're welcome!
I'm very glad I found your channel!!!!:) Thank you mam for your amazing videos.
You're welcome!
Awesome, kind of makes me want to go back to college to take a little more Ochem.
you don't have to be in college to learn. I'm learning so much since finishing schools ages ago. The learning never stops. Glad you enjoyed the video
This is the best ever ! Acetals made easy!
Thanks for the feedback! :)
this makes way more sense than in class.
I'm sorry to hear that your professor didn't do a good job explaining this, but so glad that my video was able to clarify that for you
what would the result be if a base was used? (i got asked this question)
Nice way of point to point study.. Thnku mam
You're welcome! Glad to help! :)
Thank you so much mam ! But mam, in my textbook it is written that ketones do not react with monohydric alcohols at all, and no such reaction is given. It is written that ketones react with dihydric alcohols to form cyclic ketals , and noncyclic ketals are unstable. I am still a high school student and so I do not know much about acetals and ketals.
What I think is, as cyclic ketals are stable the reaction proceeds towards right and we can isolate the product, but in case of non cyclic ketals, as they are unstable the reaction proceeds towards the reactants and we cannot isolate the product.
This is all my prediction, if I am wrong then please forgive me and clarify me.
Thank you .
I'm sorry, but I don't offer tutoring over social media. For help with questions like this and more, I recommend joining the organic chemistry study hall. Details: leah4sci.com/join or contact me through my website leah4sci.com/contact/
@@Leah4sci Oh, I am glad that atleast you replied and offered to help me mam. Thank you !
She is amazing !!!
Awww thanks!
Thank you , it helped a lot
You're welcome, glad it helped!
Asalm o alaikum...ma'm thanks for this video ...and plz make video on regeoselective and regeospecific comparison .may u live log...
That's a good idea for a video. Maybe down the line
Leah4sci thanks ma'm
could you pls make a video on transacetalization or the cascade reaction? I'm in Orgo 2 right now and we are getting into that. Unfortunately there isn't much material online regarding those processes
Thanks for the recommendation. I post free videos as I have time and am always looking for suggestions.
Hi Lea , thank you so much for your great work, I want to know are there any videos related to protecting groups in your channel to study ? or is there a way that you could advise me to study them?
I cover the Cyclic Acetal protecting group in the last video of my Acetal Series at Leah4sci.com/acetal
For more help with topics like this, I recommend joining my Organic Chemistry Study Hall. Details: leah4sci.com/join or contact me through my website leah4sci.com/contact/
Awesome!
Thanks!
You are my hero
AWWWWWW
Gracias, me ha servido de mucho
You're welcome!
You’re my savior
awwww! glad to help! :)
Thank you
God bless you
You're welcome! Glad to help! :)
You are the best
Aww thanks!
love you!!
Thanks!
this helped me a lot
Glad it helped! You're welcome :)
Thank you for the videos...they are super ¡¡¡ I have a question....why when methylating glucose for example, there is only the annomeric C which takes the CH3 and not the other OH around the other carbons of the molecule??? Thanks a lot ¡¡¡
I'm sorry, but I don't offer tutoring through TH-cam comments. For help with this and more, I recommend joining the organic chemistry study hall. Full details: leah4sci.com/join
Your express is so good. 143
Glad you like it!
Thankkkkyouuuuu
You are very welcome!
I read that ketone do not react with monohydric alcohol.. Is it true.. Clear my doubt
I'm sorry, but I don't offer tutoring over youtube comments. For help with this and more, I recommend joining the orgo study hall. Full details: leah4sci.com/join
can we not use a geminal diol to protect the carbonyl?
Thanks for watching and for the question. The standard is to use a vicinal diol as a protecting group. Using a geminal diol (where the two hydroxy groups are bound to the same carbon) causes a bit more ring strain than is desirable.
U r amazing
Thanks!
God bless u 👍
Thank you!
Love it
Glad you enjoyed it!
hellow dr.
how can i know,l solve by protect or not?
We use a protecting group if we want to perform a reaction without affecting a highly reactive group on the molecule. It is useful in synthesis when we want to react a molecule that has multiple functional groups with selectivity.
Plz, make videos for addition of cyanide ion to aldehyde and ketones
I'm currently working on other topics right now, but I will consider your suggestion for future videos :)
best :)
thanks!
Pls ur writing is covering de eqns making it hard to understand
Thanks for the feedback, I appreciate it
I love Chemistry... I want to teach chemistry for iitjee
You're very welcome
Leah. I like your videos a lot, but what I like the most, are the ridiculous comparisons. 11:04 that sentence ended me
Glad you liked it! Anything that helps it stick!