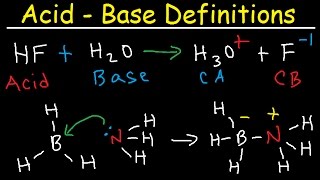
Introduction to Acids and Bases in Organic Chemistry
ฝัง
- เผยแพร่เมื่อ 20 ต.ค. 2024
- leah4sci.com/ac... presents: Introduction to Acids and Bases in Organic Chemistry including Arrhenius, Bronsted-Lowry and Lewis Explanations.
Need help with orgo? Download my free guide '10 Secrets to Acing Organic Chemistry' HERE: leah4sci.com/or...
This is the first video in my acid/base series with a focus ranking acids and bases using logic and understanding instead of calculating ka/pka values and ICE charts.
This videos gives you a logical foundation to help you understand the definitions and relationships between Arrhenius, Bronsted-Lowry and Lewis acids and bases.
Catch the entire series, along with my acid/base cheat sheet and practice prolem set on my website: leah4sci.com/ac...
For more in-depth review including practice problems and explanations, check out my online membership site: studyhall.leah4...
For private online tutoring visit my website: leah4sci.com/or...








Hi, I'm from germany and your videos are awesome. Thanks for explaining this in detail. It must be a lot of work, but i really appreciate your videos and help!
You're so welcome, happy to help! I'm so glad to hear that you enjoy my videos!
Thank you so much, I've been searching around and seeing what defines a base and a acid is, and how to predict the products for a while now.
You're very welcome!
This video was an immense help! Thank you for the clear explanation!
You're so welcome, happy to clear things up!
I love your videos! Very helpful and it goes beyond what books teach you. Please keep doing them, you're doing every organic chemistry student a favor!!
You're very welcome, happy to help!
Thank you so much, you are better at explaining the stuff more than my college professor
Happy to help!
Awesome! Thank you so much, this is the subject I am working on right now. I can't wait to share your videos with my classmates!
You're very welcome! Thanks for sharing me with your classmates :)
I like how she referred to BASE as blue in the video lol. Great video tho thanks for the help.
huh?
I read that too xD I think it's a typo lol
Remember, kids, acid and blues don't mix ;) (nahmeen ;J )
Glad you liked it and happy to help!
Nicely Explained , how to deal with acids and bases in organic chemistry. Great Work
Glad you enjoyed it!
Leah, your videos are very awesome. thank you very much
I'm glad you like them! You are very welcome!
Great video!!! on to the next one
glad you like it!
Superb Leah
Thanks!
hey leah, do you give lectures about general chemistry as well ?
I have some gen chem videos on this channel and more on my other channel th-cam.com/users/leah4scimcatvideos
its very amazing video for chemistry student thanks madam for this video
You're so welcome!
are all reactions either acid/base or redox???
No, acid/base and redox are only a couple of the different types of chemical reactions. Other categories exist, as well.
Please recommend a textbook i can get online for factors that affect acidity and basicity strength 🙏
Sorry, don't know of one.
You're 'THE BEST'
thanks!
Very nice video
You're very welcome!
Thanks 4 this,
May the force be with you Leah
:) :) :)
Anyone?!
+Leah Fisch well that's a travesty in itself!!!!!! My wife and daughter hadn't either and now theyre star wars freaks. Definitely in the order they came out with the 3 old ones from the 70's and 80's, then the brand new ones, finally the one's from early 2000's if u want to. Just watch them with all your free time ;-)
My initial reply was removed, but I have other friends telling me I need to watch it, too!
Nice job
Thank you!
OK wait so if acids are e accepting, how can they be electropositive? and if the base donates e how is it electronegative? I thought that electropositive groups tend to lose e and electronegative groups tend to gain e.
c ram . it’s electropositive (positive initially) therefore it attracts electrons. vice versa for bases.
Electropositive as the name implies, is positive or partially positive and will attract electrons. Electronegative as the name implies is negative or partially negative and so has extra electrons available to attack something positive
awesome explanation! Thank you!
You're very welcome!
thank you for the vids
You are welcome
cool, I applaude you
Thanks! Glad you liked it!
is this helpful for jee mains?
Yes, its good for your foundation
Yes, many jee students use my resources to help with their study
Good
Thanks!
thanks!!!
You're welcome!
wow... you are really the best
Thanks so much :)
i love you!
Thanks!
what is a LECTRON?!?!?!?
It's her way of remembering that the Lewis definitions deal with transfer of electrons. She made up a word lectron (a combination of lewis and electron)
thanks for explaining!
Overall, it’s fine. The audio editing is a nightmare though
Really? I don't hear it. Hope you learned something despite that!
@@Leah4sci I did, your videos are helpful
🫂
🤗
Why are you that much fast?
Do you mean I speak too fast? If so, that's how I speak, but please feel free to use the settings to slow the video down to make it better for you.