
- 8
- 208 528
Dr. McCord
เข้าร่วมเมื่อ 8 มิ.ย. 2014
Channel for Dr. McCord to put videos that relate to the chemistry classes he is teaching.
วีดีโอ
Intermolecular Forces - an introduction
มุมมอง 1.6K4 ปีที่แล้ว
Dr. McCord describes the three intermolecular forces (IMFs) that hold molecules together.
balancing a redox reaction / oxidation number method
มุมมอง 169K4 ปีที่แล้ว
Dr. McCord works through the oxidation number method of balancing a redox reaction. The reaction here is the permanganate/iron(II) reaction in acid.
Ping Pong Ball Blast Demo
มุมมอง 4085 ปีที่แล้ว
Liquid N2 in 3L soda pop bottle in trash can full of ping-pong balls. Video was shot and submitted by student Hannah Erington - thanks!
Freezing Point Depression Calculation
มุมมอง 2.2K8 ปีที่แล้ว
Dr. McCord describes a freezing point depression calculation for an aqueous solution.
First Order Kinetics problem
มุมมอง 35K9 ปีที่แล้ว
Dr. McCord shows you how to work an integrated rate law problem that follows first order kinetics. There are three parts to the problem.






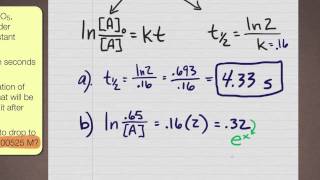
thank you sir
Wow, thank you so much!! I understand it so well now :)
Dr McCord you just balanced my grade! Thank you sir!
Was pretty helpful
You don't know how to teach 👎
thank you so much saved me for finals
where did u get the oxidation number of Mn
The oxidation no. for one oxygen is -2 ..for 4 oxygen is -8 and a negative ion above the oxygen should be added so that -8+1=-7 .....therefore mn 7+
@@Mohamed___Adhil___ oww get it, tnx
@@theyouproject welcome
This method looks very similar to the ion electron method
that was clear and extremely helpful with the steps. thank u
SOMEBODY HELP! @ 0:55 If there isn't the same number of atoms of species that are not O or H like in: N2O4 + Br^- > NO2^- + BrO3^- What do i do??? i tried just multiplying NO2^- times 2 without changing anything else including the difference in oxidation numbers (+4 - +3 = -1), and the other difference of Br being -1 - +5 = -6, i ended up having to multiply the reduction half reaction by 6, so 6N2O4 and 6(2NO2^-) = 12NO^-, but eventually the charges on the two sides did not match taking into account the protons and all
Bro/sis ....it can be easily done by the ion electron method....try it
thank you bro. you just saved my average
Thank you🙏🏿🙏🏿
How do you know which oxidation to use?
If you mean the oxidation number then you'll have to find the oxidation number of each molecule. First you'll have to learn how to find out oxidation number then you can balance chemical equations using his method .
thank you!! I finally understand and I'm so relieved I want to cry
Humble is that you..?
thanks
Well understood now
best chem guy on yt
*HE TALKS A LOT ABOUT SCIENCE STUFF PROFESSOR DAVE EXPLAINS*
Wow 🔥🔥🤩
thank you bro
Thank you very very much made it very simple 🙏🏻🙌🏻✨✨💫
Tq sir ......😊
too cool keep up the good work
Thank you!!
Thank you, sir. You helped me before my exam
thank u so much... understood the concept easily
2:29
I heard you retired, I love you man! you will go down in the hall of fame of teachers I've had in my life.
Thank you soo much sir ❤
Thank you I love u
Thank you chemistry jesus🙏🙏
GOOD VIDEO
Thank you sir❤
Nice. Thanks a lottt
what a fucking king, I love you Dr. McCord
Thankyou soooooo much. I watched many videos but not this simple thankyou once again❤
TQ so muchhhh...I wasted 30 minutes before watching this vedio..in search of this concept
Video*🤓
Nice explained
How do l know that manganese oxide is going to be decomposed to manganese 2+
It's the charge already given...in question....if not then the oxidation number of manganese metal is zero[0]
Beautiful
thank you sooo much for the best explanation ever!!
This is so awesome! I even understood when I have nothing to do with Chemistry! I got interested and was thinking, what if one of the half reaction’s change of charge is 0? I figured the 8step method does not work in that case. Then we cannot put coefficients to balance it out. Is there an easy step by step way for such a case?
If charge change would be 0,will it remain as a a redox half reaction?
the best method!!
Thankyou sir you made it so simple brilliant.
Brilliant Video
Thanks a lot
Thank you sir, really helped a lot
Thankyou so much!!!!
Thankss❤
I FINALLY UNDERSTOOD THIS, NEVER THOUGHT IT WAS THIS EASY, THANK YOU SO MUCH❤️