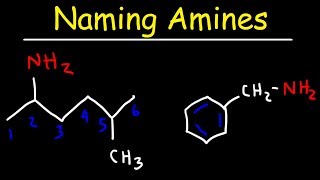
Naming Amides - IUPAC Nomenclature
ฝัง
- เผยแพร่เมื่อ 7 ก.ย. 2024
- This organic chemistry video tutorial explains how to name amides using iupac nomenclature.
Subscribe:
/ @theorganicchemistrytutor
Access to Premium Videos:
/ mathsciencetutor
/ mathsciencetutoring
New Organic Chemistry Playlist
• Organic Chemistry - Ho...








Final Exams and Video Playlists: www.video-tutor.net/
Just finished my homework in GenChem. Thanks for this😊
You're like my second lecture, thanks so much and you should make more videos on ode🙏
Not all heroes wear capes! God bless!!!
Love these videos, they’re so helpful
Powerful lesson in less than 12 min.!
you're literally saving my GPA :
saving my life for the past 4 yeras. The goat of all academic tyvm sir.
Man you are a legend thank you so much
at 6:44 wouldn't the methyl substituent on the benzene be 3-methyl ?? In all other examples you start numbering under the doubled bonded O but here the numbering started to the C attached to the benzene.
will someone answer this , I really need to no why its not 3-methyl
thought its correct 2-methyl cuz when i googled N,2-dimethybanzamide it gave the same structure !
I agree, we need someone to explain this mystery!
I don't know, but if you see naming the aromatic compounds video, you'll notice that in cases like this when ever a benzene ring is attached to a functional group, and you want to count the substitute on the ring, you start from the carbon on the ring that is attached to the functional group not from the carbon on the functional group
I don't know if I'm making sense but this is what I've noticed... Hope someone with a better understanding can help us here
yea...i don't know...someone needs to explain this plsss
The reason why it is N,2-Methylbenzamide is because the methyl is placed on the second carbon of the benzene, and not of the longest possible chain. The group's numbering is always based on its position relative to the ring, and not to the rest of the molecule. I would recommend reviewing Unsaturated hydrocarbons as it is more thoroughly explained there.
Thanks a lot. Clear and simple
You work is a piece of cake for my supper and dinner🙏
The most clutch man on youtube
Thank you for the explanation, I learnt a lot!💯
Great video. Thank you!!!
That's really helpful,my notes couldn't give me answers to some of the complex amides explained
Thanks for these, I did my degree a long long time ago...
was a little confused by the carboxamide at 5.33 (why not follow the same earlier protocol to make cyclohexanamide)?
because thats the only exception
The amide carbon is not counted in the ring
That's why it's carboxamide
Love it, u are so good. Your videos are so helpful
Thank you 💕
Thank you brotherrrrr
at 5:40 why is it cyclohexane carboxamide and not just cyclohexanamide or cyclohexan-1-amide
Because the people making the nomenclature suck and think it's funny when people forget it's written in a tough way instead of an obvious way
because amide is directly attached to the ring, its a special case.
When there is ring or symmetric structure we use carboxamide.
in aldehyde we use, carbaldehyde, etc.
hope it helped you...
the IUPAC name should be cyclohexylmethanamide
Of course it would make sense to that but you would have to realize another carbon is apart of the functional group, and because of this cyclohexanamide isn't the same as cyclohexane carboxamide
you would be surprised if you searched them both you would realize they are completely different structures. But really good observation.
Very helpful ☺️ thank you
Wow ..that was very good from you sir
you are AWESOME thank you
Thanks teacher ❤❤❤❤
Thanks:)
I understand this better than what my teacher is telling me...
thx 💕
good job sir!
So great it was!
If a tertiary amide (with ethyl and methyl as R' and R") is a substituent of an 8-carbon saturated carboxylic acid attached to its carbon #6, would you use, for example, N-ethyl-N-methyl-6-carbamoyloctanoic acid?
Thanks bro it helps a lot
Thank you
I've got a question. I saw in another video that all N- substituents should be listed before the main chain substituents. Which one is correct?
This one
Thanks a lot 😊😊
thanks alot
Sir I am in class 11th but it is still very helpful for me thanks sir
Thank u very much.
Lots of love
At 8:27 "Now let's work on a more complicated example" 😫 Wait wasn't it already getting complicated?? 😨😨😨
Lol
It was so easy example...
sahit xila xila
I’ve been writing it wrong the entire time I called it 2-Aminoethan-2-ol
Thanks
we dont need to put 1,6-
hexanediamide in the last example?
Yes it is necessary to numbering the functional group
So make it correct
what happens if there is an alcene? how does the name change
how to name it if its a substituent like how do we use its prefix in name
Carbamoyl is the prefix used for amides and make sure to not count it's carbon in principal carbon chain
5:41
I want to practice. Is there any online source where I can practice?
Great
5:49 what if Cl was in the example instead of NH2?
That becomes another function group- Acid Chloride. Name by replacing the -ic of the acid with -yl and add Chloride. In effect, that would have been benzoyl chloride.
please explain why use 'N', what is the meaning of 'N'?
it means that the alkyl group is attached to the N, it's not a ramification of the parent/principal carbon chain so it doesn't have a number associated with it
@@taniasawaya26 thank you kind stranger
Are amides always terminal?
bro didnt undersstand how that was N,2 dimethyl benzamide and not N,3 dimethyl benzamide
Benzene ring is taken as the parent chain.. not the amide one
He is right actually because it contains 3 carbonar atoms going to methyl, one C connected to N and O@@poonamramawat7637
I will dedicate my degree to you
Thanks Mark Wahlberg!
I think the naming of benzamide is wrong. You didnt take into account the carbon in the carbonyl group.
Nope it's correct
quite viiiiiiiiideo
what level is this?
10th grade in South Korea
poggers only
only poggers can like
Hello
I like my professor. But my God she over complicates this shit. Thanks man